Location:
Eng Sanderson Building, Classroom 2
Date:
Abstract
Healthcare is a major and rapidly growing sector of every developed economy, with 2015 world wide healthcare costs exceeding $7.7 trillion per annum. It is notable that US healthcare costs have risen to 18% of GDP. While the largest portion of the cost is for hospital, clinical, laboratory and physician services, some 10% is on expenditures for medicines. Despite being a modest portion of the total, the medicine component has been receiving heightened attention due, first, to its immediate impact on healthcare budgets and patient pocketbooks but also to concerns about drug safety, quality and even availability.
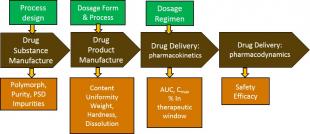
Although the process systems engineering community has had some impact on the new drug discovery process, its most notable contributions have been to facilitate improvements in product development and manufacturing. Specifically these contributions have consisted of approaches for improving the design of processes for the production of the active ingredients, the design and development of formulated drug products and the robust operation of associated manufacturing processes. In recent years, the industry and the regulatory agencies have recognized the need for manufacturing innovation, one consequence of which is the accelerating transition from batch to continuous manufacturing. This in turn has driven changes in other components of the product life cycle, from process development to supply chain management. Moreover, this openness to change has stimulated further innovations in manufacturing methods such as 3D- and drop-on-demand printing of dosage, production of film-based products, roll-to-roll printing, as well as scaled-down, point of use manufacturing systems. PSE methodologies have important roles to play in all of these innovations.
Beyond these traditional product and process focused contributions, there also have emerged opportunities to deploy process systems methodology to support enterprise-level decisions in supply chain management and product pipeline management. These applications require innovative developments in multistage stochastic optimization methodology. A further important application domain is to support clinical decisions and improve the care of patients through the individualized administration of medicine in real time or via regularly timed administration of specific dosage amounts. PSE modeling and optimization methodologies deployed in the form of automated closed loop or open-loop physician advisory systems can offer significant benefits to the patient, the clinician and the healthcare system. This bridging between the medicine and the clinical service provider points to further opportunities for PSE methodology in the healthcare services sector.
In this presentation, we will provide an overview of a selection of PSE contributions in these areas, drawing on work from the world-wide community, our collaborators and our own research group. We will outline some important research challenges that need to be addressed going forward.
Biography
Gintaras.V. (Rex) Reklaitis is Burton and Kathryn Gedge Distinguished Professor of Chemical Engineering at Purdue University (USA). He has served as Head of the School of Chemical Engineering and deputy director of the NSF Engineering Research Center on Structured Organic Particulate Systems, among other appointments. His expertise lies in process systems engineering, the application of information and computing technologies to process and product design, process operations and supply chain management. Current research interests include applications of process systems methodology to improve pharmaceutical product design, development, manufacture and administration as well as systems studies of integrated energy networks and supply chains. He was educated at the Illinois Institute of Technology (BS ChE), and received MS and PhD degrees from Stanford University. He is a member of the US National Academy of Engineering, fellow of AIChE, and past Editor-in-Chief of Computers & Chemical Engineering. Among the recognitions he has received are the Warren K Lewis and Van Antwerpen Awards of AIChE , the Pruitt Award (CCR), the Long Term Achievements in Computer Aided Process Engineering Award of the EFChE and most recently the 2017 John M Prausnitz AIChE Institute Lecturer. He has served on the Board of Directors of AICHE, the Council for Chemical Research and the CACHE Corporation and continues to serve on the editorial boards of several journals. He has published 270 papers and book chapters and edited/authored nine books.