Post date:
By Qi Zhou and Timm Krüger
Do you remember the first time you saw your own blood? Chances are that you bumped into a table as a child, or a doctor took a blood sample from your arm when you were sick. Anomalies in blood flow are associated with many common diseases, such as heart attack, stroke and malaria – diseases which threaten millions of lives globally each year.
Red blood cells (RBCs) are the most common cells in blood, occupying 40%–50% of its volume. RBCs are responsible for transporting oxygen to all organs and tissues across the human body. This is not a trivial task. During each cycle of circulation, RBCs need to travel through complex networks of blood vessels, some of which are even narrower than the RBCs themselves.
To accomplish their grand venture throughout the human body, RBCs need to be ‘deformable’ – or capable of changing their own shape to get through smaller spaces – and to share available space in blood vessels with other cells, such as white blood cells which are part of the immune system. Some diseases can cause a change in RBC deformability and the way they are arranged as they travel from one vessel to another.
Uncovering such mechanisms, especially in the smallest blood vessels with diameters less than one tenth of that of a hair, is crucial for a better understanding of the role of blood flow in health and disease. This understanding can potentially lead to better ways of diagnosing and treating blood-borne diseases, such as malaria or sickle-cell anaemia.
The direct observation of RBCs flowing in the blood vessels of humans or animals is expensive and technically difficult, but thanks to advances in microfluidics technology over the past two decades, the dynamics of RBCs can now be recreated by experiments using miniaturised devices. These devices are made up of tiny channels with bespoke geometries and well-controlled flow conditions.
Despite the increasing sophistication of such devices used to mimic blood flow for experimental purposes, they still rely on several simplifications which mean they cannot fully replicate the conditions found in real human or animal blood vessels. For example, the devices use microchannels which often have a rectangular cross-section, rather than the nearly circular cross-section of real blood vessels. In addition, experiments often use diluted blood since RBCs are difficult to distinguish when flowing at the naturally high concentrations found in the body. These technical compromises create potential uncertainties that may lead to misinterpretation of experimental results if not carefully analysed.
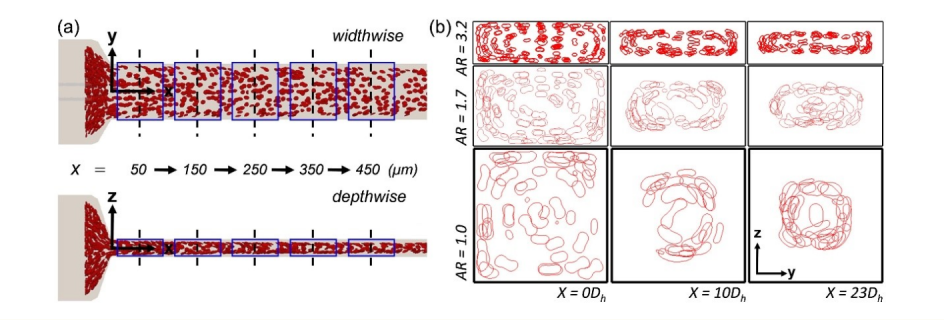
In our recent paper published in the Biophysical Journal, we reported an unexpected arrangement of RBCs when flowing through a rectangular channel such as that typically used in lab experiments. Usually, RBCs move towards the centreline of a channel or blood vessel, owing to their softness and tendency to be pushed away from the walls by fluid. However, our experimental colleagues at the University of Strathclyde in Glasgow observed that, under conditions which are often used for microfluidic experiments, RBCs can form a different spatial arrangement, where they accumulate in a ring-like structure around the centreline.
We confirmed these findings with large-scale simulations on the UK’s national supercomputer, ARCHER. Thanks to the exhaustive data generated by these simulations, we could understand and explain the unexpected RBC arrangement.
We found that the channel length required for RBCs to reach the channel centreline becomes much longer than that commonly presumed in experiments if diluted, rather than if whole blood is used. Therefore, the common assumption that RBCs are distributed close to the channel centreline may not be valid in many experiments routinely performed around the world.
Other researchers should therefore be aware of our findings to avoid misinterpretation of their data, or better still, to improve the design of their microfluidic devices.
Our work teaches us the lesson that some scientific assumptions require an occasional revision, even if they are made regularly by a large number of researchers. It seems that blood flow, although studied extensively for nearly two centuries, still has many surprises to offer.
Find out more
The article is also available on the Science Direct website.