Location:
Ashworth Labs, Lecture Theatre 3
Date:
Deracemization of Racemic Suspensions using Solution Racemization and Temperature Cycles
Abstract
Chiral molecules are vital in the natural world, and therefore find significant use as pharmaceutical and agricultural chemicals. Synthesis of chiral molecules usually produces a racemic mixture of the desired product and its counter enantiomer, a twin molecule which is the mirror image of the desired product, and with the same physical properties. This makes the separation of the two enantiomers impossible by traditional chemical engineering separations unless a chiral mediator is employed. However, the properties of the counter enantiomer in chiral environments is different to those of the desired enantiomer leading to different biological activities; this can lead to significant problems with the counter enantiomer. The disaster with the drug thalidomide is an example of this. Hence finding routes to creating pure enantiomers is essential.
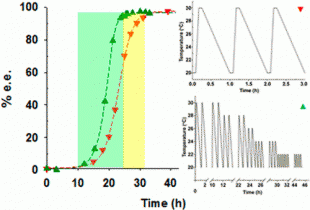
Our group recently developed a method to convert a racemic suspension of a conglomerate forming compound (where the R- and the S- crystals are distinct entities) into a suspension containing only a pure enantiomorph – thus converting the undesired enantiomer into the desired enantiomer and producing a pure product. The method requires a solution phase racemization reaction that enables conversion between the two enantiomers in solution, and temperature cycles to create periods of dissolution and crystal growth. The result is a suspension where all crystals have become one of the two pure enantiomorphs. The method is based on the method of Viedma ripening of conglomerate forming species, first demonstrated by the group of Vlieg, however has many advantages in terms of simplification of processing and scale-up. Several modifications of the temperature cycling have found to be successful.
Biography
Adrian Flood is a Professor in the Department of Chemical and Biomolecular Engineering at Vidyasirimedhi Institute of Science and Engineering in Rayong, Thailand where he is the leader of the Industrial and Pharmaceutical Crystallization group. He received a B.E. in the field of Chemical Engineering from the University of Sydney in 1990, and a Ph.D. from the University of Queensland in 1996, under the supervision of Professor Ted White. He specializes in the field of crystallization of molecular species from solution, including work on the mechanistic features of the process, experiments concerning a diverse range of molecules, including inorganic molecules, sugars, amino acids, pharmaceuticals, and proteins, and is also interested in improvements in the design of industrial crystallizers.